Home / Technology
LifeScale’s unique microfluidic sensor detects individual bacteria in each well, enabling microbe replication and culture growth to be measured with unmatched precision. But LifeScale goes further – by measuring the mass of each bacterium, it generates a detailed population profile of each culture that leads to a faster and more accurate result
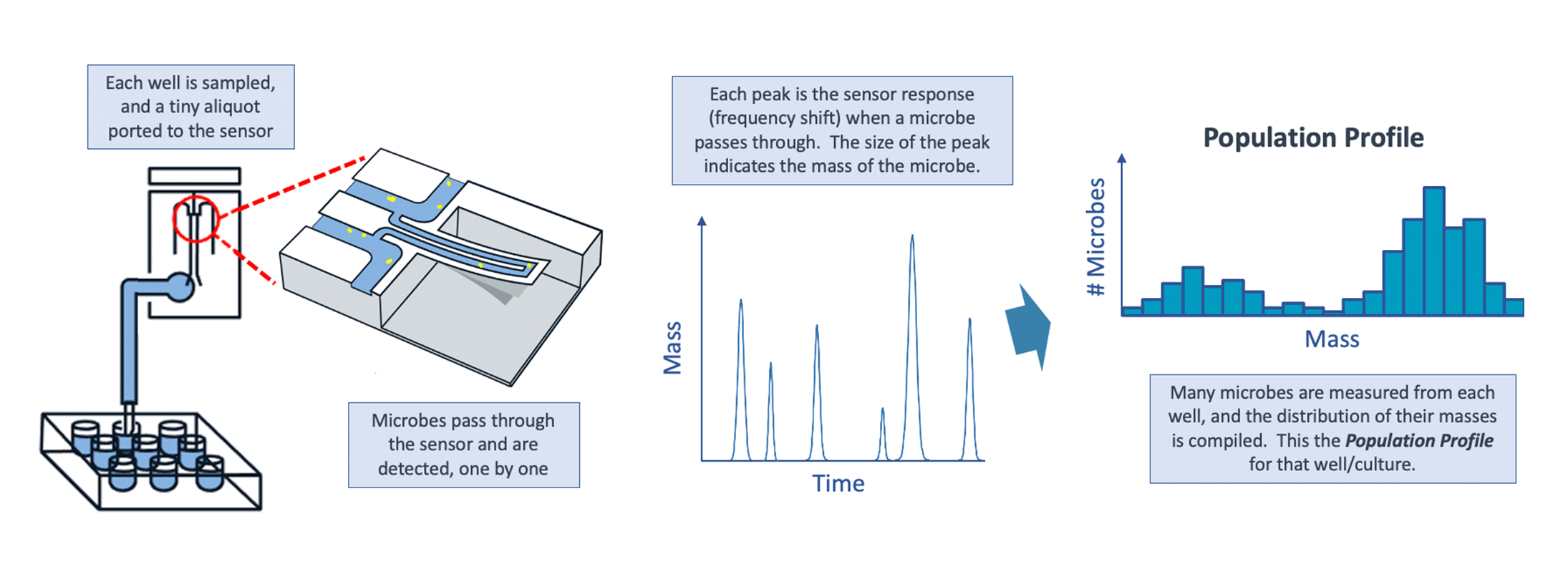
By combining the population profile data with advanced AI, LifeScale can determine correct MICs even for delayed growth phenotypes. The key is the information contained in the individual cell masses. As mentioned, delayed growth strains may not replicate for several hours. However, even during LifeScale’s short incubation time, the microbes *do* respond to the antibiotics by changing their morphology and mass, producing filamentous forms or spheroplasts. LifeScale’s population profiles clearly reveal these responses, whereas a growth-based AST can miss them since they occur prior to replication and growth of biomass.
The result is that LifeScale performance matches that of long-incubation/standard-of-care ASTs, and is superior to growth-based ones.
These results have been confirmed in a cross-platform study comparing AST performance on strains sourced from the CDC AR Bank.